
Model the patient, not the average
How subtype, stage, biomarkers, risk, and trajectory become the starting context for every therapeutic hypothesis.
Explore Topic

An AI-native biomedical initiative integrating multi-omic, clinical, and computational data to model Alzheimer's disease progression, identify biomarkers, discover therapeutic targets, and accelerate translational research through scalable scientific infrastructure.
Fuse multi-omic profiles, clinical records, literature, and computational features into one disease intelligence layer.
Map Alzheimer's progression across stages, subtypes, biomarkers, and intervention scenarios with AI-native systems biology.
Prioritize biomarkers and therapeutic targets, then move the strongest evidence into validation plans for collaborators.
The platform converts heterogeneous biomedical evidence into computable Alzheimer's disease models, then uses AI and systems biology to surface biomarkers, targets, and translational next steps.
Unifies molecular, clinical, literature, imaging, and computational signals into a shared Alzheimer intelligence layer.
Models how disease state, subtype, trajectory, and risk may evolve across preclinical and clinical stages.
Ranks candidate biomarkers, mechanisms, and targets by evidence strength, biological plausibility, and translational potential.
Packages platform findings into assays, dataset checks, collaborator workflows, and go/no-go research plans.
The project uses a therapeutic hypothesis platform to connect patient subtypes, disease stage, causal mechanisms, evidence, and intervention strategy. Each technical output becomes a testable hypothesis with expected biomarker movement, safety risks, trial implications, and a validation path.
The Alzheimer's Project is built by a multidisciplinary team where computation, biology, neuroscience, and clinical research move as one connected system.
AI researchers and ML engineers build the therapeutic hypothesis platform. Neuroscientists, molecular biologists, data scientists, physicians, and INTEC collaborators ground every platform-ranked hypothesis in real brain biology, clinical relevance, and validation discipline.
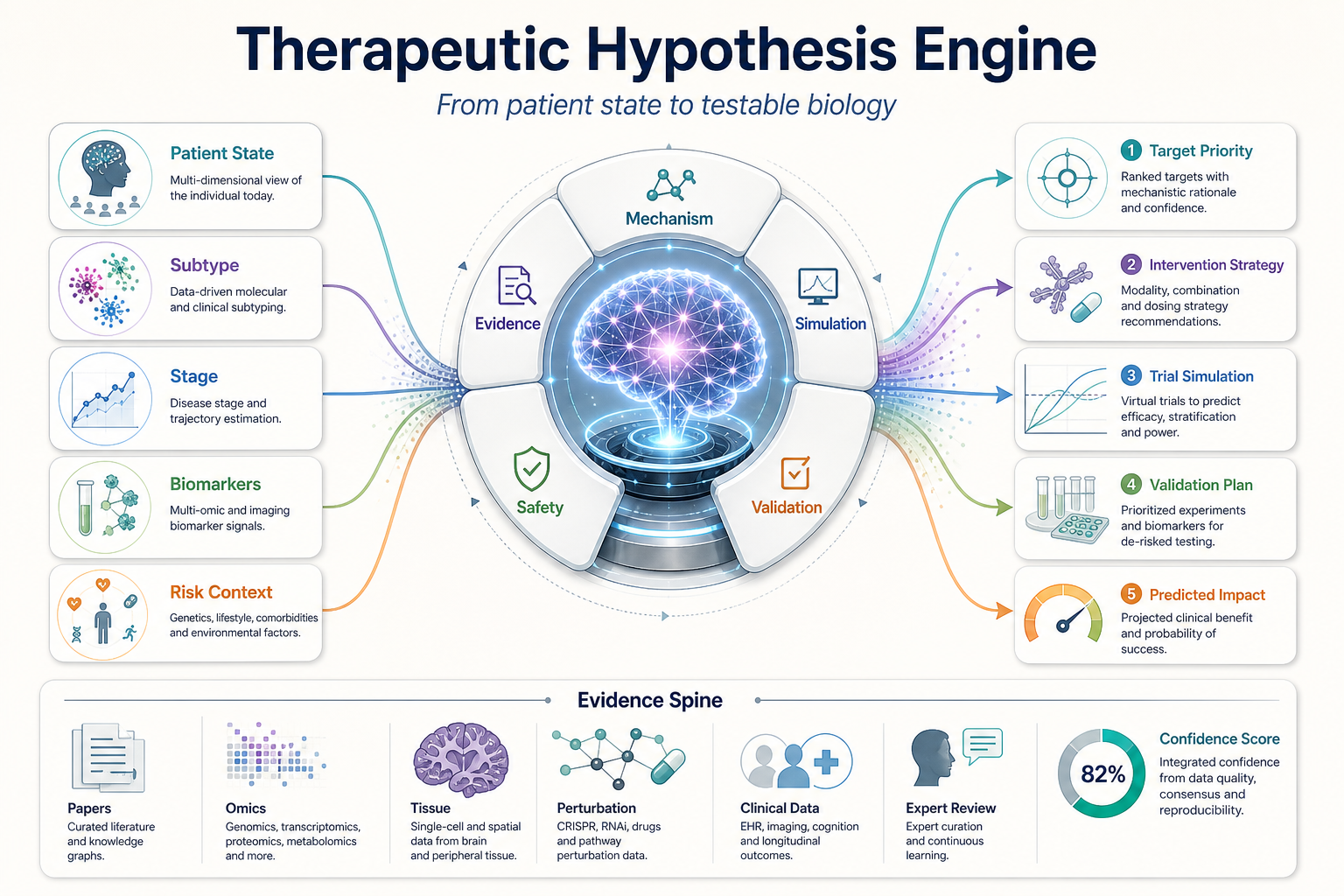


Field notes from The Alzheimer's Project: patient-state modeling, the therapeutic hypothesis object, evidence ranking, and the validation loop that turns predictions into evidence.

How subtype, stage, biomarkers, risk, and trajectory become the starting context for every therapeutic hypothesis.
Explore Topic
How a mechanism, intervention direction, expected biomarker movement, safety risk, and assay plan become one structured record.
Explore Topic
How support, contradiction, human relevance, subtype clarity, safety, and feasibility shape the next experimental priority.
Explore Topic
How tissue, perturbation, biomarkers, and clinical context define go/no-go criteria before an idea becomes a development path.
Explore Topic